
Custom Development & Manufactoring
With over 50 years in the pharmaceutical industry, we are a trusted Custom Manufacturing Organization (CMO) and Custom Development Manufacturing Organization (CDMO) for innovators and multinational pharmaceutical companies, renowned for our exceptional performance and consistently top-tier evaluations of key performance indicators (KPIs). Our dedication to quality and innovation allows us to adapt to the evolving needs of our clients and contribute significantly to the success of their products in the market.
Established Legacy
ACS DOBFAR has a rich heritage in the pharmaceutical sector, originating in Italy in 1973. This long-standing history highlights the company’s stability, experience, and continuous evolution.
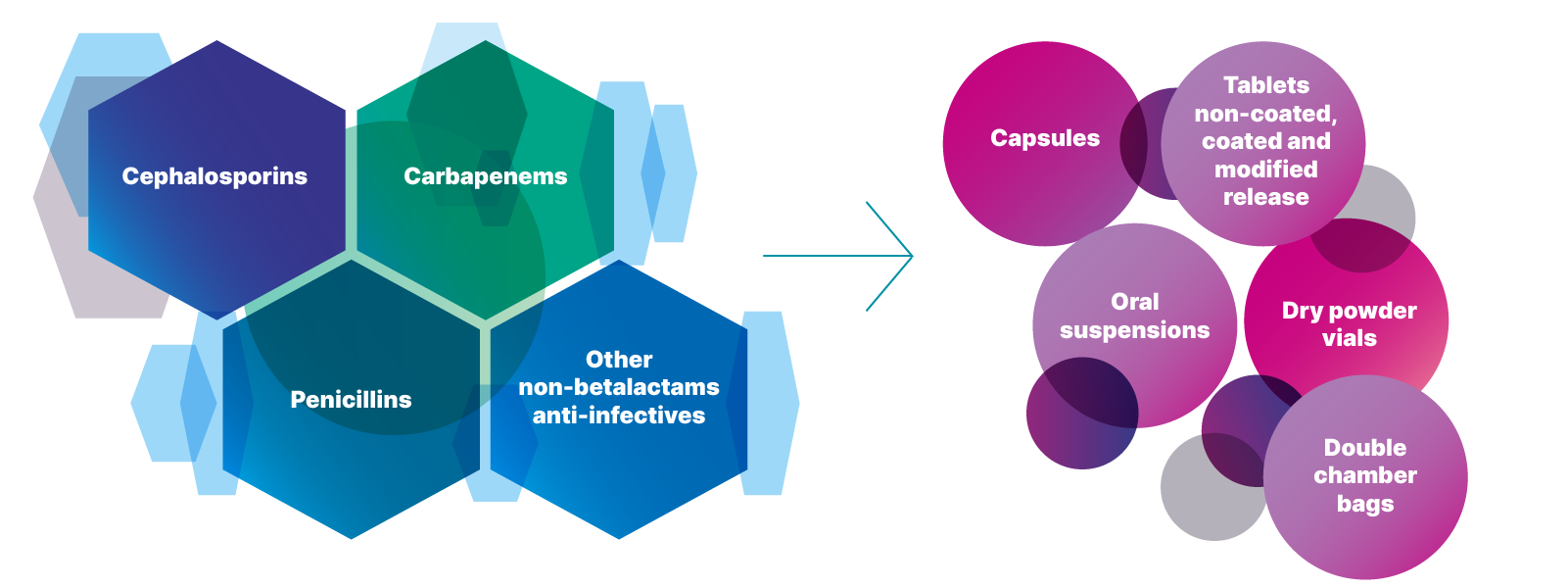
We are fully integrated manufacturer of Cephalosporin, Carbapenem and Penicillin APIs as well as Finished Dosage Formulations, both Oral and Injectable.
Operational Excellence
Our team develops tailored solutions for clients, providing custom commercial manufacturing and packaging. Our unique technologies, such as the Sterbag packaging and SmartPak delivery system, reflect our commitment to excellence.
Innovation and Technological Development
We prioritize ongoing innovation, supported by multifunctional laboratories and state-of-the-art technologies. Our continuous investment in advanced tools ensures top-quality service delivered within a competitive timeframe.
Our staff specializes in a variety of fields, ranging from analytical chemistry to fermentation and molecular biology, as well as chemical synthesis and process scale-up.
Main Activities
- Manufacturing process/analytical method improvement
- Standardized Productions for both Drug Substances and Drug Products
- Manufacturing process/analytical method development New Chemical Entity for both Drug Substances and Drug Products
- In process controls
- Stressed/Short term stability studies to support changes/new submissions
- Impurities preparation and characterization
- Analytical investigations to support Quality Control Unit and Regulatory Affairs departments
Fields of activity
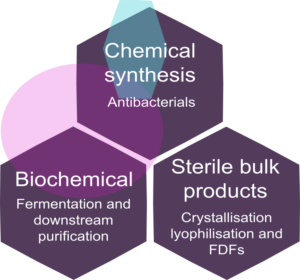
Chemical Synthesis
CHEMICAL PROCESS TECH. PERFORMED ON LAB, PILOT AND INDUSTRIAL SCALE
Alkylation
Bromination
Chlorination
Esterification and ester hydrolysis
Condensations via mixed anhydrides
Condensations via thiol-esters
Schotten-Baumann acylations
Wittig reactions
SN1 and SN2 reactions
Dieckmann condensations
Condensations via phosphorous anhydrides
Penicillin ring expansion
Homogeneous palladium catalysis (deblocking, coupling)
Silylation
Methoxylation
Catalytic hydrogenations
Vilsmaier reactions
Reformatsky reactions
Flinklestein reactions
Sulphur oxydations
Diphenydiazomethane esterification
Diazotisation
Biochemical
FERMENTATION AND DOWNSTREAM PURIFICATION
Shake flask and pilot plant fermentation
Membrane filtration
Biocatalisys process development / Enzyme immobilization
BIOTECHNOLOGIES PERFORMED ON LAB, PILOT AND INDUSTRIAL SCALE
Fermentation media and process conditions development
Harvest cells separation via centrifugation, microfiltration or ultrafiltration
Biomass homogenization / Enzyme purification
Solution concentration via ultra – membrane filtration
Solvent extraction
Enzymatic hydrolysis / Enzymatic acylations / Enzymatic oxydations
Sterile bulk products
- Bulk sterile product crystallisation (cephalosporines, penicillins, carbapenems and other) from lab up to commercial scale
- Bulk sterile product lyophilization (cephalosporines, penicillins, carbapenems and other) from lab up to commercial scale
- Dry powder filling (cephalosporines, penicillins, carbapenems):
-
- powder flowability
- vial size assessment
- demo/stability batches
Reliable partner
Solid operational background
High Performance Indicators (KPIs)
Robust partnership with Companies of Excellence